Generic Name
Composition
Therapeutic Category
Division
Linezolid
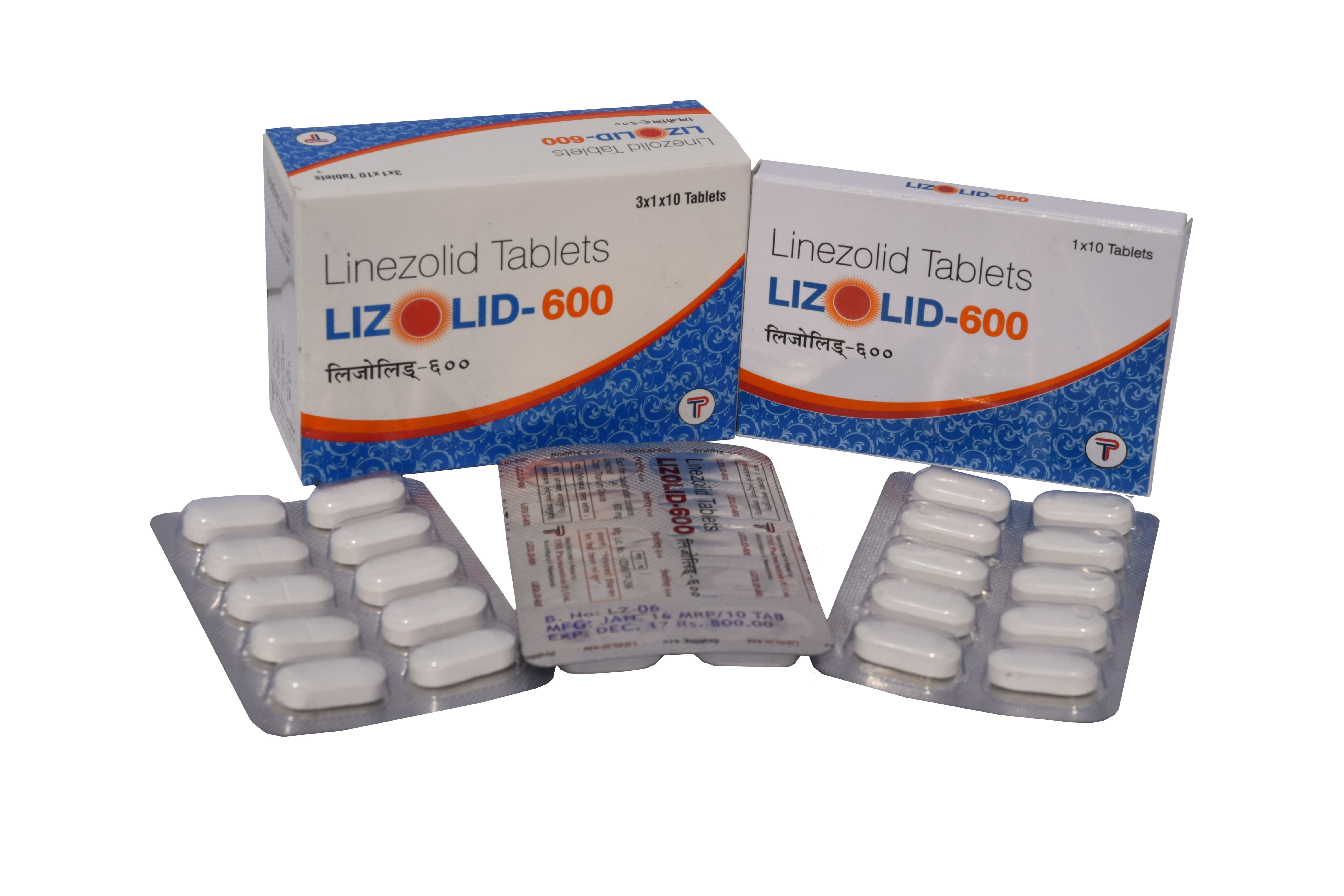
Linezolid 600mg Tablets
Oxazolidinone Antibiotic
ERRA
| BRAND | COMPOSITION | DESCRIPTION |
|---|---|---|
| Linezolid 600 mg | Each film coated tablet contains: Linezolid 600mg |
White, oblong, biconvex, film coated tablet with breakline on one side |
PHARMACOLOGY
Pharmacotherapeutic Group:oxazolidinone
Pharmacodynamic Properties:Linezolid is an bacteriostatic oxazolidinone, inhibiting ribosomal protein synthesis at the level of the 23s ribosome of the 50s subunit. It is active against gram-positive bacteria including vancomycin-resistant enterococci and MRSA. It has limited in-vitro activity against gram-negative bacteria.
Pharmacokinetic Properties:
Absorption: Oral bioavaiability: Approximately 100%. Rapidly absorbed after oral admin. Peak plasma concentrations after 1-2 hours, although delayed if administered with high fat meal.
Distribution: Plasma protein binding: Approximately 31%. Readily distributed into well perfused tissues, bone, fat, lungs, muscle, skin blister fluids and CSF. Volume of distribution at steady state: 40-50 liter.
Metabolism: Oxidised to 2 main inactive metabolites.
Excretion: Approximately 65% of dose eliminated via non renal route. Renal clearance is low. Elimination half life: 4.6-7 hours (oral).
INDICATIONS
Nosocomial Pneumonia, Community acquired pneumonia, Complicated and Uncomplicated skin and skin structure Infections, Vancomycin resistant Enterococcus Faecium Infecions
Dosage and Administration
Adult: 600 mg oral every 12 hours
ADVERSE DRUG REACTIONS
Diarrhoea (antibiotic associated colitis reported), headache, nausea, vomiting, constipation, abnormal liver function tests, fever, vaginal and oral candidiasis, skin rash, pruritus, dizziness, insomnia, anaemia, tongue discoloration, taste disturbance, lactic acidosis, optic and peripheral neuropathy (particularly if used ≥ 28 days).
Potentially Fatal: Reversible myelosuppression including anaemia, leukopenia, pancytopenia and thrombocytopenia (particularly if using > 10-14 days), transient ischaemic attacks, renal failure, Stevens-Johnson syndrome.
PRECAUTIONS
Preexisting myelosuppression, renal impairment (CrCl < 30ml/min), uncontrolled hypertension, phaeochromocytoma, carcinoid syndrome, untreated hyperthyroidism, chronic infection, history of seizures, bipolar depression, schizophrenia or acute confusional states. Pregnancy and lactation. Monitor complete blood counts weekly. Give after haemodialysis. Not known if linezolid or metabolites removed during peritoneal dialysis.
DRUG INTERACTIONS
May reduce serum level with rifampin and phenytoin. May cause hypoglycemia with insulin or oral antidiabetics. May increase risk of seizures with tramadol. Increased risk of serotonin syndrome with Monoamine oxidase inhibitors (MAOIs), Tricyclic Antidepressants (TCAs), Serotonin-norepinephrine reuptake inhibitors (SNRIs), or other serotonergic drugs (e.g. bupropion, vilazodone, mirtazapine, meperidine, trazodone, nefazodone). Significant increase in blood pressure with vasopressive agents (e.g. epinephrine, norepinephrine), sympathomimetic agents (e.g. pseudoephrine) & dopaminergic agents (e.g. dopamine, dobutamine)
CONTRAINDICATIONS
Patients with a known allergy to linezolid. Patients taking any medicinal product which inhibits monoamine oxidases A or B (e.g., phenelzine, isocarboxazid) or within two weeks of taking any such medicinal product.
PRESENTATION
LIZOLID: Each box contains 3 Blister of 10 Tablets per strip
PATIENT INFORMATION |
|---|
|
Food Interaction: Beverages and foods high in tyramine content may cause hypertensive crisis. Avoid dietary tyramine ≥100 mg per meal. Administration: May be taken with or without food. Pregnancy: Category C Storage: Store at 25°C. Protect from light. Missed dose: Take the missed dose as soon as you remember. Skip the missed dose if it is almost time for your next scheduled dose. Do not take extra medicine to make up the missed dose |