Generic Name
Composition
Therapeutic Category
Division
Fexofenadine HCl
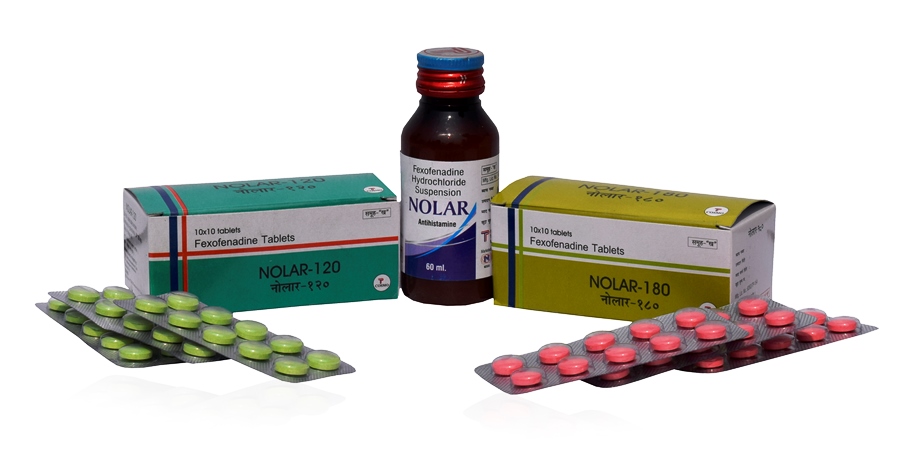
Fexofenadine HCl 120mg Tablets
Fexofenadine HCl 180mg Tablets
Fexofenadine HCl 30mg/5ml Suspension
Anti-histaminic/Anti-allergic
COSMO
| BRAND | COMPOSITION | DESCRIPTION |
|---|---|---|
| NOLAR 120 | Each film coated tablet contains: Fexofenadine Hydrochloride 120mg |
Yellow colored, oblong, film coated tablet with breakline on one side. |
| NOLAR 180 | Each film coated tablet contains: Fexofenadine Hydrochloride 180mg |
Brick red colored, oblong, biconvex, film coated tablet with breakline on one side. |
| NOLAR SUSPENSION | Each 5ml contains: Fexofenadine Hydrochloride BP 30mg |
White colored suspension filled in amber coloured PET bottle. Each filled bottle is labelled and packed in carton. |
Pharmacotherapeutic Group: Anti-allergic agent, H1 receptor antagonist
Pharmacodynamic properties: NOLAR (Fexofenadine), is a competitive peripheral histamine (H1) receptor antagonist that acts on effector cells in the GI tract, blood vessels and respiratory tract. It works by blocking histamine (H1) receptors and thus prevents activation of H1 receptor-containing cells by histamine. Therefore, it help to alleviate symptoms such as runny nose and sneezing; red, watery, or itchy eyes; itchy nose, throat or roof of the mouth; itchy skin and rash. NOLAR causes less drowsiness than other antihistamines.
Pharmacokinetic properties:
Absorption: Absorbed rapidly from the GI tract;Time to peak plasma concentrations: 2-3 hr.
Distribution: Enters breast milk. Protein binding: 60-70%; Does not readily pass through the blood-brain barrier.
Metabolism: Approximately 5% of the total dose eliminated by hepatic metabolism.
Excretion: Mainly via faeces, via urine (10% of dose); Elimination half-life: 14 hours; May be prolonged in renal impairment.
Allergic rhinitis; Chronic urticaria
Seasonal allergic rhinitis: 120 mg once daily.
Chronic idiopathic urticaria: 180 mg once daily.
Child: 2-11 yr: 30 mg (5ml) twice daily.
6 month -2 yr: 5mg (2.5ml) twice daily.
PATIENT INFORMATION |
|---|
|
Administration:
Food Interaction: Fruit juices such as grapefruit, orange and apple may reduce the bioavailability and exposure of NOLAR. Pregnancy: Category C Nursing Mothers: There are no adequate and well controlled studies in women during lactation. However, when terfenadine was administered to nursing mothers, fexofenadine was found to cross into human breast milk. Therefore, NOLAR is not recommended for breast-feeding women. Storage: Store in a cool and dry place; Protect from light, heat and moisture; Keep away from the reach of children.
|
Hypersensitivity
Renal and hepatic impairment; Pregnancy;lactation; Heart disease; Medication that lead to fast or irregular heart disease; Children
Viral infection (cold/flu); headache, dizziness, drowsiness, fatigue; nausea, dyspepsia, dysmenorrhoea.
Co-administration with ketoconazole or erythromycin may increase plasma levels of fexofenadine; Administration of antacid containing aluminium or magnesiuhydroxide causes reduction in bioavailability; Pramlintide may increase the anticholinergic effect of fexofenadine; Bioavailability may be increased by verapamil; Efficacy may be reduced by rifampin; May increase adverse effects of other anticholinergics and CNS depressants; May increase arrhythmogenic effect of antipsychotic agents (phenothiazines); avoid concurrent usage; May reduce the efficacy of betahistine.
NOLAR 120: Each box contains 10 blister packs of 10 tablets per blister.
NOLAR 180: Each box contains 10 blister packs of 10 tablets per blister.
NOLAR Suspension: Each bottle contains 60ml of suspension.